Education:
Ph.D.: 1982, Kansas State University
Awards and Honors:
George Lynn Cross Professor
National Institute of Health (NIAID) Merit Award
Fellow, American Academy of Microbiology
President’s Associates Presidential Professor, University of Oklahoma Health Sciences Center
Endowed Professor, University of Oklahoma Health Sciences Center
Pre-OUHSC:
University of California at Los Angeles
Research Interests:
Structural biology and biochemistry of bacterial protein toxins
Teaching:
Basic Bacteriology
Contact Information:
Office: BRC 309
Email: Rodney-Tweten@ouhsc.edu
Research Interests:
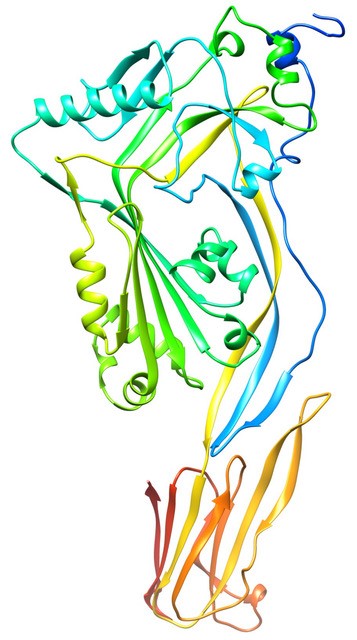
Studies on the Cholesterol-dependent Cytolysins (CDCs). Our research centers on the study of the mechanism of pore-forming toxins and proteins from prokaryotic and eukaryotic sources, although we have recently expanded into studies that involve an uncharacterized anti-bacterial toxin produced by certain species of the colonic microbiome. Over the past 30+ years our studies have primarily focused on the pore-forming mechanisms of a variety of toxins produced by a wide variety of pathogenic bacteria with an emphasis on the cholesterol dependent cytolysins (CDCs). More than 100 bacteria species carry a gene for a CDC and they have been shown to play significant roles in the pathogenesis of such bacterial species as Clostridium perfringens (gas gangrene), Listeria monocytogenes (listeriosis), Streptococcus pneumoniae (pneumoniae, meningitis) and Streptococcus pyogenes (strep throat, necrotizing fasciitis). The study of the CDC structure and pore-forming has led to our understanding of how human immune defense proteins, such as the complement membrane attack complex and perforin (MACPF) family of proteins, as well as a large class of protein toxins from poisonous marine fish, form their pores in the cell membrane An intriguing characteristic of these proteins is the ability of these proteins to make the transition from a soluble state to a homo-oligomeric pore-forming complex on the membrane. The mechanism by which they accomplish this transition is complex and each protein family contributes unique aspects to this process. Although we have worked extensively on the CDC mechanism there still remain several intriguing aspects of their mechanisms that remain unknown. We employ a variety of biochemical and biophysical approaches to the study of the CDC mechanism.
Studies on the CDC-like pore-forming proteins. Our studies of the CDCs have recently led to our discovery of a large family of CDC-like pore-forming toxins (CDCLs), the genes of which are present in >300 species of bacteria that span ecological niches from the Saharan desert to the Arctic and Antarctic waters and are found in numerous important species of the human oral and colonic microbiomes. Our initial studies indicate that they are distant relatives of the CDCs and their targets are unknown, as their binding domains are evolutionary distinct from that of the CDCs, as well as each other. Furthermore, many of these CDCLs from the human colonic microbiome species, as well as the gut microbiomes of insects, have evolved what appears to be a two component pore complex, which is distinct from that of the CDCs. Hence, the study of these CDC-like proteins is just beginning and will likely lead to a new frontier of study to understand how these proteins assemble a pore and their cellular targets.
Bacteroidetes anti-bacterial toxin. Our last project also involves a new field of study of, thus far, a unique antibacterial toxin produced by species of Bacteroides and Prevotella present in the human gut and oral microbiomes. This study is a collaboration with Dr. Laurie Comstock (University of Chicago). The mechanism of this toxin remains unknown and it has no analogs that suggest how it functions to kill or arrest microbial growth. We have determined that two proteolytic cleavage sites must be cleaved in sequence to activate the toxin and have identified its receptor on susceptible cells. Our future studies will be primarily focused on determining its structure, the mechanism of its activation and the mechanism by which it kills/inhibits susceptible bacterial species.
Current Lab Personnel:
Casie Collamore, Ph.D.
Tristan Sanford, Graduate Student
Hunter Abrahamsen, Graduate Student
Patricia Parrish, Research Technician
Selected Publications:
- Czajkowsky, D. M., E. M. Hotze, Z. Shao, and R. K. Tweten. 2004. Vertical collapse of a cytolysin prepore moves its transmembrane β-hairpins to the membrane. EMBO J 23:3206-3215.
- Giddings, K. S., J. Zhao, P. J. Sims, and R. K. Tweten. 2004. Human CD59 is a receptor for the cholesterol-dependent cytolysin intermedilysin. Nat Struct Mol Biol 11:1173-1178.
- Ramachandran, R., R. K. Tweten, and A. E. Johnson. 2004. Membrane-dependent conformational changes initiate cholesterol-dependent cytolysin oligomerization and intersubunit β-strand alignment. Nat Struct Mol Biol 11:697-705.
- Polekhina, G., K. S. Giddings, R. K. Tweten, and M. W. Parker. 2005. Insights into the action of the superfamily of cholesterol-dependent cytolysins from studies of intermedilysin. Proc Natl Acad Sci 102:600-605.
- Ramachandran, R., R. K. Tweten, and A. E. Johnson. 2005. The domains of a cholesterol-dependent cytolysin undergo a major FRET-detected rearrangement during pore formation. Proc Natl Acad Sci 102:7139-7144.
- Schuerch, D. W., E. M. Wilson-Kubalek, and R. K. Tweten. 2005. Molecular basis of Listeriolysin O pH-dependence. Proc Natl Acad Sci 102:12537–12542
- Soltani, C. E., E. M. Hotze, A. E. Johnson, and R. K. Tweten. 2007. Structural elements of the cholesterol-dependent cytolysins that are responsible for their cholesterol-sensitive membrane interactions. Proc Natl Acad Sci U S A 104:20226-20231.
- LaChapelle, S., R. K. Tweten, and E. M. Hotze. 2009. Intermedilysin-receptor interactions during assembly of the pore complex: assembly intermediates increase host cell susceptibility to complement-mediated lysis. J Biol Chem 284:12719-12726.
- Farrand, A. J., S. LaChapelle, E. M. Hotze, A. E. Johnson, and R. K. Tweten. 2010. Only two amino acids are essential for cytolytic toxin recognition of cholesterol at the membrane surface. Proc Natl Acad Sci U S A 107:4341-4346.
- Wickham, S. E., E. M. Hotze, A. J. Farrand, G. Polekhina, T. L. Nero, S. Tomlinson, M. W. Parker, and R. K. Tweten. 2011. Mapping the intermedilysin-human CD59 receptor interface reveals a deep correspondence with the binding site on CD59 for complement binding proteins C8alpha and C9. J Biol Chem 286:20952-20962.
- Dowd, K. J., A. J. Farrand, and R. K. Tweten. 2012. The cholesterol-dependent cytolysin signature motif: a critical element in the allosteric pathway that couples membrane binding to pore assembly. PLoS Pathog 8:e1002787.
- Dunstone, M. A., and R. K. Tweten. 2012. Packing a punch: the mechanism of pore formation by cholesterol dependent cytolysins and membrane attack complex/perforin-like proteins. Curr Opin Struct Biol 22:342-349.
- Hotze, E. M., E. Wilson-Kubalek, A. J. Farrand, L. Bentsen, M. W. Parker, A. E. Johnson, and R. K. Tweten. 2012. Monomer-monomer interactions propagate structural transitions necessary for pore formation by the cholesterol-dependent cytolysins. J Biol Chem 287:24534-24543.
- Wade KR, Hotze EM, Briles DE, Tweten RK. 2014. Mouse, but not human, ApoB-100 lipoprotein cholesterol is a potent innate inhibitor of Streptococcus pneumoniae pneumolysin. PLoS Pathog 10:e1004353.
- Farrand AJ, Hotze EM, Sato TK, Wade KR, Wimley WC, Johnson AE, Tweten RK. 2015. The cholesterol-dependent cytolysin membrane-binding interface discriminates lipid environments of cholesterol to support beta-barrel pore Insertion. J Biol Chem 290:17733-17744.
- Lukoyanova N, Kondos SC, Farabella I, Law RH, Reboul CF, Caradoc-Davies TT, Spicer BA, Kleifeld O, Traore DA, Ekkel SM, Voskoboinik I, Trapani JA, Hatfaludi T, Oliver K, Hotze EM, Tweten RK, Whisstock JC, Topf M, Saibil HR, Dunstone MA. 2015. Conformational changes during pore formation by the perforin-related protein pleurotolysin. PLoS Biol 13:e1002049.
- Wade KR, Hotze EM, Kuiper MJ, Morton CJ, Parker MW, Tweten RK. 2015. An intermolecular electrostatic interaction controls the prepore-to-pore transition in a cholesterol-dependent cytolysin. Proc Natl Acad Sci U S A 112:2204-2209.
- Lawrence SL, Gorman MA, Feil SC, Mulhern TD, Kuiper MJ, Ratner AJ, Tweten RK, Morton CJ, Parker MW. 2016. Structural Basis for Receptor Recognition by the Human CD59-Responsive Cholesterol-Dependent Cytolysins. Structure 24:1488-98.
- Christie MP, Johnstone BA, Tweten RK, Parker MW, Morton CJ. 2018. Cholesterol-dependent cytolysins: from water-soluble state to membrane pore. Biophys Rev 10:1337-1348.
- Wade KR, Lawrence SL, Farrand AJ, Hotze EM, Kuiper MJ, Gorman MA, Christie MP, Panjikar S, Morton CJ, Parker MW, Tweten RK. 2019. The structural basis for a transition state that regulates pore formation in a bacterial toxin. mBio 10:e00538-19.
- Burns, JR, Morton, CJ, Parker, MW and Tweten, RK. 2019. An intermolecular π-stacking interaction drives conformational changes necessary to β-barrel formation in a pore-forming toxin. mBio 10:e01017-19
- Evans, J. C., Johnstone, B. A., Lawrence, S. L., Morton, C. J., Christie, M. P., Parker, M. W., and Tweten, R. K. 2020. A Key motif in the cholesterol-dependent cytolysins reveals a large family of related proteins. mBio 11
- Evans, J.C., McEneany, Valentina Laclare, Coyne, Michael J., Caldwell, Elizabeth P., Sheahan, Madeline L., Von, Salena S., Coyne, Emily M., Tweten, Rodney K., Comstock, Laurie E. 2022. A proteolytically activated antimicrobial toxin encoded on a mobile plasmid of Bacteroidales induces a protective response. Nat Commun 2022; 13:4258.
- See all publicaations at https://www.ncbi.nlm.nih.gov/pubmed/?term=Tweten+R